(-)items
-
Medical Supplies
-
Cleaning & Sanitary
Adhesives, Sealants & Tapes
Air Freshener & Odor
Air & HVAC Filters
Bathroom Tissue & Dispensers
Brooms & Dustpans
Brushes
Carts, Trucks, Storage
Cleaning Chemicals
Chemicals, Lubricants & Paints
Facility Maintenance
First Aid & Safety
Floor & Carpet Care
Floor Care Equipment
Gloves
Hand Sanitizers
Hand Wipers & Rags
- Clinical Laboratory
-
Apparel
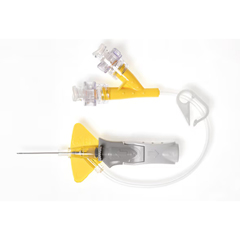
BD Nexiva® Closed IV Catheter 24 Gauge
Double MarkdownPrice is viewable only to staff. Log inPlease Sign In or Create an Account
Sign In
Create Account
We're sorry, this item is out of stock. We're sorry, this item is currently unavailable. Get notifiedGet Notified
Free Shipping!In StockRx RequiredNexiva® Closed IV Catheter 24 Gauge
The Nexiva Closed IV Catheter 24 Gauge is a sterile, disposable closed IV catheter with a winged hub and Q-Syte luer access split septum. It fits IV access workflows where buyers want a catheter built for stability and comfort, along with needle technology that helps confirm vessel entry during insertion. This 24 gauge version pairs a Vialon biomaterial catheter with a stainless steel safety needle and a 1.2 mm extension tube inner diameter.
Good Fit For
- IV access setups needing a closed catheter design
- Facilities standardizing on winged hub catheters with safety needle protection
- Clinical workflows where insertion confirmation is important
Key Features
- 24 gauge closed IV catheter for IV access procedures
- Soft, flexible stabilization platform is designed for stability, comfort, and reduced catheter movement in the vessel
- Instaflash Needle Technology helps confirm vessel entry to support more focused insertion
- Passive needle-shielding engages as the needle is withdrawn for added safety handling
- Q-Syte luer access split septum port supports closed catheter access
- Sterile, disposable design with Vialon biomaterial catheter and stainless steel safety needle
Applications and Usage
- Peripheral IV catheter insertion
- Closed IV access workflows using a split septum port
- Single-use sterile catheter placement in clinical settings
Packaging and Handling
- Packaging: EA.
- Size: 1.2 mm.
- Material: Plastic.
- Sterility: Sterile.
- This SKU is a 24 gauge catheter, so gauge selection should match the intended IV access protocol.
- The port type is Q-Syte luer access split septum; buyers should confirm it matches their preferred access setup.
- The catheter uses a winged hub and integrated stabilization platform, which may matter for clinicians who prefer that handling style.
FAQ
- What type of IV product is this? This is a closed IV catheter in 24 gauge.
- Does this catheter include a safety needle feature? Yes. The Nexiva closed IV catheter uses a safety needle, and passive needle-shielding engages as the needle is withdrawn.
- What port type does this catheter use? This catheter uses a Q-Syte luer access split septum port.
- Is the catheter sterile and disposable? Yes. This catheter is sterile and intended for disposable use.
- What helps with catheter stability during use? The catheter is integrated with a soft, flexible stabilization platform designed for stability, comfort, and to minimize catheter movement in the vessel.
From Nexiva by BD.
- HCPCS : A4351
- Hub Type : Winged
- Sterile : Yes
- Extension Tube Inner Diameter : 1.2 mm
- Port Type : Q-Syte* Luer Access Split Septum
- Safety Feature : Safety Needle
- Application : Closed IV Catheter
- Usage : Disposable
- Color : Yellow
- Needle Material : Stainless Steel
- Catheter Material : Vialon Biomaterial
- Gauge : 24 Gauge
- Brand : Nexiva.
- Catheter Length : 3/4"
- Hub Material : Plastic
- UNSPSC Code : 42221504
- UNSPSC Title : Intravenous or arterial cannulas or accessories
Prescription Information
A valid medical prescription must be submitted to Betty Mills within seven (7) days for the ordered item(s), and shipping will be on hold until the prescription is received. If the prescription is not provided within this timeframe, the order may be canceled. This requirement does not apply to licensed care providers, training facilities, equivalent qualified professionals, or licensed wholesalers, who are exempt from this policy.
Prescriptions may be sent via:
Fax(650) 443-5201EmailMailThe Betty Mills Company, Inc.
19 South B Street Suite 8
San Mateo, CA 94401You are entitled to a free consultation following the purchase of this item.
Sizing Chart
Product Documents
Needles & Syringes Top Sellers
-
Infusion Pump Fanny Pouch, 1/EA (MON 1140266EA)
$29.39 Each
Customers also Viewed
This weeks Deals
-
Prevent® Sharps Container (MON 855063EA)
$4.45 Each
Prescription Required
A valid medical prescription must be submitted to Betty Mills within seven (7) days for the ordered item(s), and shipping will be on hold until the prescription is received. If the prescription is not provided within this timeframe, the order may be canceled. This requirement does not apply to licensed care providers, training facilities, equivalent qualified professionals, or licensed wholesalers, who are exempt from this policy.